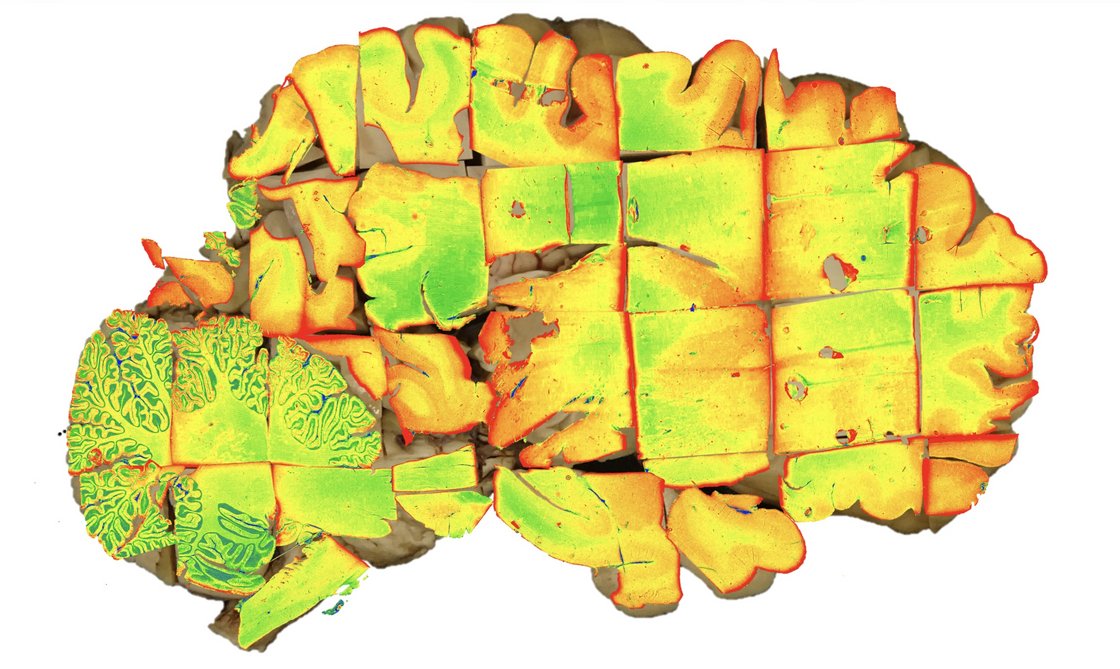
Prof Friederike Liesche-Starnecker's team combines modern neuropathology with innovative digital analysis methods and translational research.
A particular focus here is on computer-aided (neuro)pathology. The combination of classic histopathological diagnostics with digital and AI-based methods opens up new possibilities for the objective and reproducible evaluation of complex image data.
A central thematic focus is on human bornavirus encephalitis, a rare but so far almost always fatal and therefore increasingly relevant zoonotic disease. The aim of our research is to better understand the immunological and pathogenetic mechanisms of this neurotropic viral infection. In particular, we are investigating processes of virus spread, host-virus interaction and immune-mediated damage to the nervous system.
The team is also involved in the field of translational neuro-oncology. The focus here is on the biological basis of aggressive brain tumours, their interaction with the tumour environment and the breakdown of tumour heterogeneity. By closely linking research and clinical application, we aim to contribute to the faster transfer of innovative findings into patient care.
On this page you will find further information on our research priorities and current projects. We are always happy to exchange ideas with interested partners and to enter into new scientific collaborations. We also welcome enquiries for collaboration in the context of final theses or doctorates.
Main Research Areas
Immunology and Pathogenesis of Human Bornavirus Encephalitis
Borna disease virus 1 (BoDV-1) is a rare neurotropic virus that can cause severe and often fatal encephalitis in humans. For a long time, it was only known from veterinary medicine, particularly in horses and sheep. In 2018 it has been proven that BoDV-1 also affects humans. The virus is mainly found in Bavaria and is currently considered untreatable.
The working group has been researching the Borna virus for several years, with a focus on the immunological and pathogenetic characterisation of Borna disease virus 1 (BoDV-1). The aim is to understand basic mechanisms of virus entry, spread in the nervous system and interaction with the host immune response. VSBV-1, another zoonotic Bornavirus with human pathogenicity, is being investigated comparatively.
A central focus is the immunopathology of human bornavirus encephalitis, especially in comparison to animal infection, and on the characterisation of microglia and CNS-associated macrophages. Spatially resolved transcriptomic, proteomic and imaging methods are used to analyse virus-infected cells, local immune responses and dissemination pathways of BoDV-1 in the central and peripheral nervous system.
Due to their model character, these studies provide fundamental insights into neurotropic viral infections and immune-mediated diseases of the nervous system.
ZooBoFo Bavaria 2.0 - Bornavirus in the One Health Context
In the "Zoonotic Bornavirus Focalpoint Bavaria 2.0" (ZooBoFo Bavaria 2.0) project, we are pursuing a consistent One-Health approach in order to better understand transmission routes of the virus and further develop prevention strategies. The project combines interdisciplinary expertise from human and veterinary medicine, environmental research and neuropathology. Key components include reservoir studies in shrews, autopsy-based clinical research and the further development of diagnostic and antiviral approaches.
ZooBoFo Bavaria 2.0 is coordinated by the Bavarian State Office for Health and Food Safety (LGL) and is being developed in close cooperation with the University Hospital Regensburg, the Friedrich Loeffler Institute (FLI) and other partners. The project isfundedby the Bavarian State Ministry of Health, Care and Prevention.
INSIGHT - Microglia and Immune Responses in Bornavirus Encephalitis
In the INSIGHT (Investigating Microglia Signatures in Human Tissue) project, we are investigating how the brain's own immune system, in particular microglia and CNS-associated macrophages, react to a Bornavirus infection. The aim is to identify disease-specific immune cell signatures in human tissue and to gain new insights into the pathogenesis of neurotropic viral infections.
INSIGHT is being developed in close cooperation with Dr. Jana Koch from the Cellular Neuroanatomy group at the Department of Anatomy and Cell Biology at the University of Augsburg (Prof. Dr Peter Wieghofer), who has particular expertise in microglia and macrophage biology.
Jungbäck N, Grochowski P, Hieber D, Dinser M, Mielewczyk Z, Pfefferkorn T, Muntau B, Richter T, Rieder G, Bayas A, Hirschbühl K, Märkl B, Adam P, Tappe D, Liesche-Starnecker F. Multimodal profiling of immune responses reveals innate-adaptive immune imbalance in human bornavirus encephalitis. Acta Neuropathol Commun 14, 109 (2026). doi: 10.1186/s40478-026-02319-6.
Michelakaki E, Schade B, Boehm B, Kappe E, Suchowski M, Kupca A, Schumacher M, Gager AM, Liesche-Starnecker F, Fiedler S, Schwarz E, Bago Z, Blutke A, Beer M, Rubbenstroth D, Matiasek K. Borna disease virus 1 as cause of fatal meningoencephalomyelitis in wild hedgehogs, Germany, 2022–2025. Emerg Infect Dis (2026). doi: 10.3201/eid3205.250952.
Vollmuth Y, Jungbäck N, Grochowski P, Mögele T, Stark L, Zarrabi NS, Schlegel J, Schaller T, Märkl B, Matiasek K, Liesche-Starnecker F. Mapping Bornavirus encephalitis-A comparative study of viral spread and immune response in human and animal dead-end hosts. PLoS Pathog 21, 8 (2025). doi: 10.1371/journal.ppat.1013400. PMID: 40758738. PMCID: PMC12338802.
Jungbäck N, Vollmuth Y, Mögele T, Grochowski P, Schlegel J, Schaller T, Märkl B, Herden C, Matiasek K, Tappe D, Liesche-Starnecker F. Neuropathology, pathomechanism, and transmission in zoonotic Borna disease virus 1 infection: a systematic review. Lancet Infect Dis. 25, 4 (2025). doi: 10.1016/S1473-3099(24)00675-3. PMID: 39793593.
Ebinger A, Santos PD, Pfaff F, Dürrwald R, Kolodziejek J, Schlottau K, Ruf V, Liesche-Starnecker F, Ensser A, Korn K, Ulrich R, Fürstenau J, Matiasek K, Hansmann F, Seuberlich T, Nobach D, Müller M, Neubauer-Juric A, Suchowski M, Bauswein M, Niller HH, Schmidt B, Tappe D, Cadar D, Homeier-Bachmann T, Haring VC, Pörtner K, Frank C, Mundhenk L, Hoffmann B, Herms J, Baumgärtner W, Nowotny N, Schlegel J, Ulrich RG, Beer M, Rubbenstroth D. Lethal Borna disease virus 1 infections of humans and animals - in-depth molecular epidemiology and phylogeography. Nat Commun. 15, 7908 (2024). doi: 10.1038/s41467-024-52192-x. PMID: 39256401. PMCID: PMC11387626.
Bayas A, Menacher M, Lapa C, Tappe D, Maurer C, Liesche-Starnecker F, Schneider H, Naumann M. 18fluorodeoxyglucose PET/CT as possible early diagnostic tool preceding MRI changes in Borna disease virus 1 encephalitis. Lancet. 403, 10427 (2024). doi: 10.1016/S0140-6736(24)00049-7. PMID: 38368015.
Vollmuth Y, Jungbäck N, Mögele T, Schmidt-Graf F, Wunderlich S, Schimmel M, Rothe C, Stark L, Schlegel J, Rieder G, Richter T, Schaller T, Tappe D, Märkl B, Matiasek K, Liesche-Starnecker F. Comparative study of virus and lymphocyte distribution with clinical data suggests early high dose immunosuppression as potential key factor for the therapy of patients with BoDV-1 infection. Emerg Microbes & Infections 13 (2024). doi: 10.1080/22221751.2024.2350168; PMID: 38687703. PMCID: PMC11107860.
Liesche-Starnecker F, Schifferer M, Schlegel J, Vollmuth Y, Rubbenstroth D, Delbridge C, Gempt J, Lorenzl S, Schnurbus L, Misgeld T, Rosati M, Beer M, Matiasek K, Wunderlich S, Finck T. Hemorrhagic lesion with detection of infected endothelial cells in human bornavirus encephalitis. Acta Neuropathol. 144, 2 (2022). doi: 10.1007/s00401-022-02442-3. PMID: 35657496. PMCID: PMC9164175.
Applied Informatics for Medical Research
Computer-assisted neuropathology deals with the development and application of information technology methods for analysing neuropathological data. The focus of this research area is to support the other research areas through the systematic and automated analysis of extensive data sets. In this way, efficient and reproducible handling of diagnostic, pathogenetic and translational questions is enabled.
The methodological focus is currently on the automated and interactive evaluation of digital histological preparations (whole slide imaging). To this end, methods from image processing, machine learning and artificial intelligence are used to segment tissue structures, characterise cell populations and identify disease-specific patterns.
Future projects will expand the focus to include the integration of multimodal data. Histological findings will be linked with molecular, transcriptomic and clinical information in order to better understand complex disease mechanisms and, for example, identify new biomarkers. In particular, computer-aided analysis enables the investigation of spatial correlations in tissue and their relationship to clinical progression.
In addition to the strongly applied, analysis-orientated research, methodological questions of machine learning and artificial intelligence are also dealt with in the domain of neuropathology.
As a cross-sectional area, computer-assisted neuropathology supports all of the Institute's research fields and enables new questions to be asked through the systematic use of digital data. The work contributes to establishing digital pathology as an integral part of neuropathological diagnostics and research.
CuBATS - Computer Based Antigen Target Selection
CAR T-cell therapy is an emerging personalised cancer treatment. However, its success depends crucially on identifying suitable target antigens that reliably occur on tumour cells and spare healthy tissue as much as possible. In glioblastoma in particular, this selection is extremely challenging due to the high degree of heterogeneity.
CuBATS addresses this problem from the perspective of computer-assisted (neuro)pathology: target antigens are determined individually for each patient in a reproducible and explainable manner using immunohistochemistry and image processing algorithms. This allows expression patterns to be analysed directly in the tissue context and their spatial heterogeneity to be visualised. Instead of just looking at the presence of a marker, this approach allows an assessment of the actual tumour specificity in the tissue - a key prerequisite for safe CAR-T therapies.
CUBATS is being developed in close cooperation with the Department of Neurosurgery at the University Hospital of Würzburg. The project is technically supported by the DigiHealth Institute at Neu Ulm University of Applied Sciences and the University of Würzburg.
Krauss S, Spieß E, Hieber D, Schobel J, Kramer F, Müller D. Overcoming Domain Shift in Atypical Mitotic Figure Detection with Deep Ensemble Learning.Studies in Health Technology and Informatics, 336 (2026). doi: 10.3233/SHTI260112.
Bücheler V, Hieber D, Karthan M, Jungbäck N, Dinser M, Pohl C, Pryss R, Liesche-Starnecker F, Schobel J. Eye-Tracking in Digital Pathology: A Vendor-Agnostic Platform for Standardized and Reproducible Eye-Tracking Studies, IEEE 38th International Symposium on Computer-Based Medical Systems (CBMS) (2025), doi: 10.1109/CBMS65348.2025.00190.
Hieber D, Haisch N, Grambow G, Holl F, Liesche-Starnecker F, Pryss R, Schlegel J, Schobel J. Comparing nnU-Net and deepflash2 for Histopathological Tumor Segmentation. Stud Health Technol Inform. 316 (2024). doi: 10.3233/SHTI240487. PMID: 39176815.
Transnational Neurooncology: Tumour Heterogeneity & Microenvironment
In the field of translational neurooncology, a central focus of research at the Institute of Neuropathology is the intratumoural heterogeneity of glioblastoma, which is regarded as an essential basis for therapy resistance and rapid tumour progression in this disease.
Our work aims to systematically characterise the spatial and cellular diversity within tumours and link it to clinical progression. By combining classical neuropathological methods with modern molecular and digital analysis approaches, we investigate how different tumour cell populations, microenvironment components and immunological signatures contribute to the dynamics and adaptation of glioblastomas.
Another translational focus is the development of innovative diagnostic strategies for better tumour characterisation and therapy monitoring. This includes, in particular, liquid biopsy approaches based on cerebrospinal fluid samples, which we have used, for example, to detect cytokine signatures after intraoperative radiotherapy of brain metastases.
These approaches help to translate the complex biology of malignant brain tumours into diagnostically useful parameters and open up new avenues for personalised therapy monitoring and treatment.
ORIGIN - Glioblastoma Heterogeneity and Resistance to Therapy
A central focus of research at our institute is the question of why glioblastoma recurs so frequently despite intensive therapy. A key driver is the pronounced intratumoural heterogeneity: different tumour regions differ morphologically, epigenetically and in their sensitivity to therapy, which favours the development of resistance.
In the ORIGIN project (Overcoming therapy resistance caused by intratumour heterogeneity in glioblastoma by utilising spatially obtained organoids), we are developing an innovative translational platform in which patient-specific organoid models are generated from several spatially separated tumour areas of a glioblastoma. These make it possible to systematically record regional differences in the tumour and test their sensitivity ex vivo to standard therapies such as temozolomide and new targeted substances.
The project is being carried out in close collaboration with the Würzburg neurosurgeon Dr Vera Nickl, whose working group has extensive expertise in the establishment and automated generation of patient-derived glioblastoma organoids. Our aim is not only to describe heterogeneity, but also to use it as a basis for individualised therapeutic approaches and to develop new strategies for overcoming resistance to therapy.
Shiban E, Krauss P, Schaller T, Konietzko I, Mögele T, Jungbäck N, Mielke D, Märkl B, Kahl KH, Mielewczyk Z, Liesche-Starnecker F. A prospective observational study of inflammatory mediators in cerebrospinal fluid after intraoperative radiotherapy of brain tumors. Ther Adv Med Oncol. 17 (2025). doi: 10.1177/17588359251389740. PMID: 41262211. PMCID: PMC12623645.
Wagner A, Brielmaier MC, Kampf C, Baumgart L, Aftahy AK, Meyer HS, Kehl V, Höhne J, Schebesch KM, Schmidt NO, Zoubaa S, Riemenschneider MJ, Ratliff M, Enders F, von Deimling A, Liesche-Starnecker F, Delbridge C, Schlegel J, Meyer B, Gempt J. Fluorescein-stained confocal laser endomicroscopy versus conventional frozen section for intraoperative histopathological assessment of intracranial tumors. Neuro Oncol. 26, 5 (2024). doi: 10.1093/neuonc/noae006. PMID: 38243410. PMCID: PMC11066924.
Godbole S, Voß H, Gocke A, Schlumbohm S, Schumann Y, Peng B, Mynarek M, Rutkowski S, Dottermusch M, Dorostkar MM, Korshunov A, Mair T, Pfister SM, Kwiatkowski M, Hotze M, Neumann P, Hartmann C, Weis J, Liesche-Starnecker F, Guan Y, Moritz M, Siebels B, Struve N, Schlüter H, Schüller U, Krisp C, Neumann JE. Multiomic profiling of medulloblastoma reveals subtype-specific targetable alterations at the proteome and N-glycan level. Nat Commun. 15, 6237 (2024). doi: 10.1038/s41467-024-50554-z. PMID: 39043693. PMCID: PMC11266559.
Prokop G, Wiestler B, Hieber D, Withake F, Mayer K, Gempt J, Delbridge C, Schmidt-Graf F, Pfarr N, Märkl B, Schlegel J, Liesche-Starnecker F. Multiscale quantification of morphological heterogeneity with creation of a predictor of longer survival in glioblastoma. Int J Cancer. 153, 9 (2023). doi: 10.1002/ijc.34665. PMID: 37501565.
Publications
Krauss S, Spieß E, Hieber D, Schobel J, Kramer F, Müller D. Overcoming Domain Shift in Atypical Mitotic Figure Detection with Deep Ensemble Learning.Studies in Health Technology and Informatics, 336 (2026). doi: 10.3233/SHTI260112.
Jungbäck N, Grochowski P, Hieber D, Dinser M, Mielewczyk Z, Pfefferkorn T, Muntau B, Richter T, Rieder G, Bayas A, Hirschbühl K, Märkl B, Adam P, Tappe D, Liesche-Starnecker F. Multimodal profiling of immune responses reveals innate-adaptive immune imbalance in human bornavirus encephalitis. Acta Neuropathol Commun 14, 109 (2026). doi: 10.1186/s40478-026-02319-6.
Michelakaki E, Schade B, Boehm B, Kappe E, Suchowski M, Kupca A, Schumacher M, Gager AM, Liesche-Starnecker F, Fiedler S, Schwarz E, Bago Z, Blutke A, Beer M, Rubbenstroth D, Matiasek K. Borna disease virus 1 as cause of fatal meningoencephalomyelitis in wild hedgehogs, Germany, 2022–2025. Emerg Infect Dis (2026). doi: 10.3201/eid3205.250952.
Liesche-Starnecker F, Mielewczyk Z, Grochowski P, Jungbäck N, Adam P, Märkl B, Schlegel J. Hirnsektion in der Allgemeinpathologie. Pathologie (2026).doi: 10.1007/s00292-026-01543-0.
Reitsam NG, Gäble A, Siebenhüter L, Schaller T, Liesche-Starnecker F, Sipos E, Dintner S, Walz C, Babic J, Trepel M, Kircher M, Fincke VE, Johann PD, Märkl B, Lapa C, Enke JS. FAP expression as a marker of malignant transformation enabling in vivo characterization in peripheral nerve sheath tumors: a multimodal and translational study. Acta Neuropathol 151, 11 (2026). doi: 10.1007/s00401-026-02979-7. PMID: 41591566. PMCID: PMC12847211.
Here you will find a selection of publications prior to the foundation of the institute. Further publications can be found on the detailed pages of the contact persons for the individual research areas.
Shiban E, Krauss P, Schaller T, Konietzko I, Mögele T, Jungbäck N, Mielke D, Märkl B, Kahl KH, Mielewczyk Z, Liesche-Starnecker F. A prospective observational study of inflammatory mediators in cerebrospinal fluid after intraoperative radiotherapy of brain tumors. Ther Adv Med Oncol. 17 (2025). doi: 10.1177/17588359251389740. PMID: 41262211. PMCID: PMC12623645.
Bücheler V, Hieber D, Karthan M, Jungbäck N, Dinser M, Pohl C, Pryss R, Liesche-Starnecker F, Schobel J. Eye-Tracking in Digital Pathology: A Vendor-Agnostic Platform for Standardized and Reproducible Eye-Tracking Studies, IEEE 38th International Symposium on Computer-Based Medical Systems (CBMS) (2025), doi: 10.1109/CBMS65348.2025.00190.
Vollmuth Y, Jungbäck N, Grochowski P, Mögele T, Stark L, Zarrabi NS, Schlegel J, Schaller T, Märkl B, Matiasek K, Liesche-Starnecker F. Mapping Bornavirus encephalitis-A comparative study of viral spread and immune response in human and animal dead-end hosts. PLoS Pathog 21, 8 (2025). doi: 10.1371/journal.ppat.1013400. PMID: 40758738. PMCID: PMC12338802.
Jungbäck N, Vollmuth Y, Mögele T, Grochowski P, Schlegel J, Schaller T, Märkl B, Herden C, Matiasek K, Tappe D, Liesche-Starnecker F. Neuropathology, pathomechanism, and transmission in zoonotic Borna disease virus 1 infection: a systematic review. Lancet Infect Dis. 25, 4 (2025). doi: 10.1016/S1473-3099(24)00675-3. PMID: 39793593.
Wagner A, Brielmaier MC, Kampf C, Baumgart L, Aftahy AK, Meyer HS, Kehl V, Höhne J, Schebesch KM, Schmidt NO, Zoubaa S, Riemenschneider MJ, Ratliff M, Enders F, von Deimling A, Liesche-Starnecker F, Delbridge C, Schlegel J, Meyer B, Gempt J. Fluorescein-stained confocal laser endomicroscopy versus conventional frozen section for intraoperative histopathological assessment of intracranial tumors. Neuro Oncol. 26, 5 (2024). doi: 10.1093/neuonc/noae006. PMID: 38243410. PMCID: PMC11066924.
Godbole S, Voß H, Gocke A, Schlumbohm S, Schumann Y, Peng B, Mynarek M, Rutkowski S, Dottermusch M, Dorostkar MM, Korshunov A, Mair T, Pfister SM, Kwiatkowski M, Hotze M, Neumann P, Hartmann C, Weis J, Liesche-Starnecker F, Guan Y, Moritz M, Siebels B, Struve N, Schlüter H, Schüller U, Krisp C, Neumann JE. Multiomic profiling of medulloblastoma reveals subtype-specific targetable alterations at the proteome and N-glycan level. Nat Commun. 15, 6237 (2024). doi: 10.1038/s41467-024-50554-z. PMID: 39043693. PMCID: PMC11266559.
Ebinger A, Santos PD, Pfaff F, Dürrwald R, Kolodziejek J, Schlottau K, Ruf V, Liesche-Starnecker F, Ensser A, Korn K, Ulrich R, Fürstenau J, Matiasek K, Hansmann F, Seuberlich T, Nobach D, Müller M, Neubauer-Juric A, Suchowski M, Bauswein M, Niller HH, Schmidt B, Tappe D, Cadar D, Homeier-Bachmann T, Haring VC, Pörtner K, Frank C, Mundhenk L, Hoffmann B, Herms J, Baumgärtner W, Nowotny N, Schlegel J, Ulrich RG, Beer M, Rubbenstroth D. Lethal Borna disease virus 1 infections of humans and animals - in-depth molecular epidemiology and phylogeography. Nat Commun. 15, 7908 (2024). doi: 10.1038/s41467-024-52192-x. PMID: 39256401. PMCID: PMC11387626.
Bayas A, Menacher M, Lapa C, Tappe D, Maurer C, Liesche-Starnecker F, Schneider H, Naumann M. 18fluorodeoxyglucose PET/CT as possible early diagnostic tool preceding MRI changes in Borna disease virus 1 encephalitis. Lancet. 403, 10427 (2024). doi: 10.1016/S0140-6736(24)00049-7. PMID: 38368015.
Hieber D, Haisch N, Grambow G, Holl F, Liesche-Starnecker F, Pryss R, Schlegel J, Schobel J. Comparing nnU-Net and deepflash2 for Histopathological Tumor Segmentation. Stud Health Technol Inform. 316 (2024). doi: 10.3233/SHTI240487. PMID: 39176815.
Vollmuth Y, Jungbäck N, Mögele T, Schmidt-Graf F, Wunderlich S, Schimmel M, Rothe C, Stark L, Schlegel J, Rieder G, Richter T, Schaller T, Tappe D, Märkl B, Matiasek K, Liesche-Starnecker F. Comparative study of virus and lymphocyte distribution with clinical data suggests early high dose immunosuppression as potential key factor for the therapy of patients with BoDV-1 infection. Emerg Microbes & Infections 13 (2024). doi: 10.1080/22221751.2024.2350168; PMID: 38687703. PMCID: PMC11107860.
Prokop G, Wiestler B, Hieber D, Withake F, Mayer K, Gempt J, Delbridge C, Schmidt-Graf F, Pfarr N, Märkl B, Schlegel J, Liesche-Starnecker F. Multiscale quantification of morphological heterogeneity with creation of a predictor of longer survival in glioblastoma. Int J Cancer. 153, 9 (2023). doi: 10.1002/ijc.34665. PMID: 37501565.
Liesche-Starnecker F, Schifferer M, Schlegel J, Vollmuth Y, Rubbenstroth D, Delbridge C, Gempt J, Lorenzl S, Schnurbus L, Misgeld T, Rosati M, Beer M, Matiasek K, Wunderlich S, Finck T. Hemorrhagic lesion with detection of infected endothelial cells in human bornavirus encephalitis. Acta Neuropathol. 144, 2 (2022). doi: 10.1007/s00401-022-02442-3. PMID: 35657496. PMCID: PMC9164175.