The heart is the first organ to emerge and function in the mammalian embryo - a remarkable feat of biological engineering that begins only days after life itself. Its formation requires the precise orchestration of highly dynamic processes: specification of cardiac progenitor cells, their migration to the nascent heart field, differentiation into specialized cardiac cell types, intricate signaling within and between tissues, complex morphogenetic remodeling, and ultimately growth into a fully functional organ. Even subtle disturbances in these tightly coordinated events can lead to severe congenital heart defects.
While the anatomical development of the heart has been studied for decades in exquisite detail, the molecular and cellular mechanisms driving cardiogenesis remain far from fully understood. Deciphering the genetic programs and signaling networks that govern heart development is therefore of immense importance - not only for developmental biology, but also for understanding the origins of congenital and acquired cardiovascular disease, which continue to represent a major clinical challenge from prenatal life through adulthood.
At the core of our research group, Molecular Cardiology, lies the ambition to uncover these fundamental mechanisms of heart formation, function, and regeneration. Using cutting-edge in vitro systems, including iPSC-derived cardiomyocytes and advanced cell culture models, alongside powerful in vivo models such as zebrafish and mouse, we investigate the genetic, molecular, and cellular programs that control cardiac lineage specification, differentiation, maturation, growth, and repair.
Our long-term vision is to decode the complex regulatory networks that shape the developing and adult heart - and to translate this knowledge into innovative therapeutic strategies for inherited and acquired heart disease.
Research

Nexilin, is a cardiomyopathy-associated F-actin binding protein, which is expressed specifically in human heart and skeletal muscle. Located at the Z-disks of sarcomeric units, it has an essential role in the maintenance of Z-disks and sarcomere integrity. Identified functions of Nexilin are to protect and stabilize Z-disks from forces generated within muscle contraction. Dilated cardiomyopathy-causing gene variants of Nexilin seem to lose the attachment ability to the sarcomeric scaffolding complex and hence Z-disk alignments become disrupted. To understand the molecular composition of cardiac Z-disks regarding how Nexilin is integrated, we are searching for new protein binding partners.Besides the structural character, Nexilin is also analyzed for its potential functional impact in biochemical signalling, mechanosensation and mechanotransduction.
The development of the vertebrate heart is not only dependent on molecular specifications and morphogenetic movements but also on organ growth. Cardiac growth can be accomplished by either hyperplasia or hypertrophy. Early in embryonic heart development cardiac growth is mainly due to hyperplasic cardiomyocyte proliferation hereby increasing cardiac cell mass. Thereafter the heart enlarges for the most part by hypertrophic expansion of the cardiomyocyte cell volume. The analysis of zebrafish mutants with defects in heart growth can help sheding light onto the molecular mechanisms beneath hyperplasic and hypertrophic processes.
In humans, cardiac infarction leads to myocardial cell death and scar formation, which subsequently lead to heart insufficiency. The zebrafish however has the capability to regenerate the heart completely after myocardial damage. Therefore we aim to understand the molecular pathways orchestrating myocardial regeneration and proliferation in zebrafish to develop therapeutic treatment strategies.
In search for molecules that control cardiac contractility, we isolated the zebrafish mutant main squeeze (msq), which displays progressive heart failure due to a mutation (msqL308P) within the Integrin-linked kinase (ILK) gene. It has been shown that the ILK-Pinch-Parvin (IPP) complex is crucial for mechanotransduction in cardiomyocytes by controlling the expression of the stretch responsive genes anf and vegf via PKB signaling (Bendig et al. 2006). For a better understanding of mechanotransduction in cardiomyocytes it is essential to learn more about the underlying molecular mechanisms. Therefore we investigate known interactors of ILK, by morpholino antisense oligonucleotide mediated gene knockdown in zebrafish.
Another aim is to identify new compounds which are able to reconstitute the cardiac function in msq mutants. In cooperation with the Karlsruher Institute of technology (K.I.T), we developed a high-troughput, fully-automated bioanalytics screening platform (Spomer et al., 2012). This robotics-assisted multicamera microscope allows us to easily screen the therapeutic relevance of small compound libraries on our zebrafish mutants in 96- or 384 well formats.
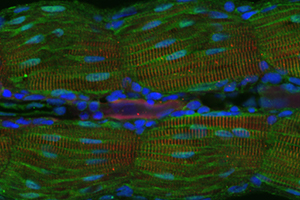
Myofibrillar myopathies (MFM) are progressive diseases of human heart and skeletal muscle. They are often associated with premature death. The muscle pathology is characterized by desmin-positve protein aggregates and myofibrillar degeneration. Half of all MFM are caused by mutations in genes encoding sarcomeric and extra-sarcomeric proteins (desmin, filamin C, plectin, VCP, FHL1, ZASP, myotilin, and αB-crystallin). Other half of these diseases are due to so far unidentified gene defects. The onset of the progressive muscle symptoms varies depending on the affected gene. FHL1- and plectin-related MFM may manifest either in childhood, adolescence or adulthood. Desmin- and αB-crystallin-related MFM tend to manifest in early and middle adulthood. Whereas disease onset beyond the forth decade of life is typical for myotilin-, ZASP-, VCP and filamin C-gene mutations. Also the clinical value is variable in MFM. Skeletal muscle weakness in the lower extremities is the most frequent initial clinical symptom. Additionally certain patterns of extra skeletal muscle involvement are typical for subtypes of MFM. Cardiac pathology is a characteristic hallmark of MFM due to desmin, αB-crystallin, FHL1 gene mutations. To date, no causative or even ameliorating therapies exist for these numerically significant group of hereditary myopathies. Since the molecular pathomechanism are largely unknown, a good model organism is essential for the identification and further analysis of the suitable illness genes. The obtained findings can later serve as a basis for the development of new therapies.
Some hereditary diseases are fatal for the patients but they are still not completely clarified. One of those is Epidermolysis bullosa simplex with muscular dystrophy (MD-EBS). It is characterized by severe skin blistering associated with muscular involvement and caused by defects in the plectin gene. Plectin is expressed in nearly all mammalian cells and acts as cytoskeletal crosslinker between microfilaments, microtubules and intermediate filaments.
With the aid of transgenic fish lines and knockdowns via morpholino injections, we are trying to characterize Plectin and its isoforms to achieve a better understanding of plectin and related diseases.
By positional cloning we identified a nonsense mutation within the SET- and MYND-domain containing protein 1 gene (smyd1) to be responsible for the flatline phenotype which shows disturbed sarcomere assembly restricted to fast-twitch skeletal muscle and heart muscle leading to loss of contraction. The methyltransferase Smyd1 localizes to the M-line, where it associates with myosin, and the nucleus, where it regulates gene expression. By rescue experiments several human mutation variants were tested because of their relevance of possibly leading to dilatative cardiomyopathies in humans.
Team
- Head of Workgroup
- Office
- Postdoctoral Medical Scientists
- Physician Scientists
- PhD Students
- Technicians
- Fish Facility
- Former Members

Prof. Dr. med. Mirjam Keßler
Schwerpunkte
Fachärztin für Innere Medizin und Kardiologie
Zusatzqualifikation Interventionelle Kardiologie
Zusatzbezeichnung Intensivmedizin
Zusatzqualifikation Herzinsuffizienz
Zusatzqualifikation Sportmedizin (Stufe 1)
- Interventionelle AV-Klappentherapie, invasive und nicht-invasive Herzinsuffizienztherapie

Prof. Dr. med. Karolina Weinmann-Emhardt
Leiterin Ablative Elektrophysiologie
Schwerpunkte
Fachärztin für Innere Medizin und Kardiologie
Zusatzbezeichnung Intensivmedizin
Zusatzqualifikation Spezielle Rhythmologie - Invasive Elektrophysiologie und Aktive Herzrhythmusimplantate
- Invasive und nicht-invasive Elektrophysiologie

Irina Lengard

Helge Stranz

Gerhard Bückle
Dr. Janessa Hofeichner
Dr. Alena Boos
Dr. Amelia Glazier
Dr. Kevin Thiessen
Dr. Isabelle Schneider
Dr. Christoph Paone
Dr. Diana Krattenmacher
Dr. Maryam Shahid
Dr. Monika Kustermann, geb. Forster
Dr. Linda Raphel
Dr. Julia Segert
Dr. Steven Rudeck
Dr. Sofia Hirth
Dr. Muzamil Khan
Dr. John Bührdel
Dr. Daniela Asam
Dr. Linda Manta, geb. Gärtner
Dr. Eva Patzel
Dr. Ina Berger
Technicians
Renate Durst
Regine Baur
Hedwig Frank
Kristin Haugg
Publications
Park DD, Kim S, Boos A, Andrasch Y, Krieg L, Rottbauer W and Just S.Smarca4 maintains mitochondrial homeostasis and energy metabolism during cardiac development. Cell Mol Life Sci. 2026;83.
Park DD, Boos A, Hermann A, Kim S, Rottbauer W and Just S.Smarce1-dependent modulation of Stat3 signaling governs cardiomyocyte proliferation. Biol Res. 2026;59.
Kim S, Park DD, Glazier A, Rottbauer W and Just S.WASHC3 knockout disrupts mitochondrial protein homeostasis and energy metabolism in cardiomyocytes. Frontiers in cardiovascular medicine. 2026;13:1682381.
Hoffmann S, Diebold S, Roeth R, Lowen A, Mellein S, Just S and Rappold GA. Linking shox/shox2 deficiency with fgfr3 gain-of-function and natriuretic peptides. Front Endocrinol (Lausanne). 2026;17:1803846.
Diofano F, Madac IEG, Koeble D, Rottbauer W, Weinmann-Emhardt K and Just S.BAG3(V468M) impairs proteasomal protein clearance and induces dilated cardiomyopathy in vivo. Biochem Biophys Res Commun. 2026;821:153900.
Buhler A, Aigner-Radakovics K, Diofano F, Marchi E, Weinmann-Emhardt K, Paquay A, Lilja SV, Rosensteiner B, Harpell R, Bernard E, Just S, Bittner RE, Lyon GJ and Kustermann M. Phenotypic variability in female individuals with the NAA10 missense variants p.(L126R), p.(L126V), or p.(F128L) leading to NAA10-related syndrome. Mol Cell Pediatr. 2026;13.
Radecke K, Rheinert D, Lowen A, Wiedmann F, Diebold S, Diofano F, Weiss B, Roth R, Schmitteckert S, Clauss S, Kaab S, Just S, Schmidt C, Rappold GA and Hoffmann S. CAV1-A Susceptibility Gene for Atrial Fibrillation: The Impact of Coding and Noncoding Variants. Journal of the American Heart Association. 2026:e041586.
Diofano, F., Amadi, C., Hartmann, L., Gahr, B.M., Weinmann-Emhardt, K., Rottbauer, W., and Just, S. (2025). SMYD1-mediated mono-methylation of lysine K35 of sarcomeric myosin heavy chain (MHC) regulates sarcomere assembly and homeostasis in zebrafish and human iPSC-derived cardiomyocytes. J Mol Cell Cardiol 208, 74-84.
Park, D.D., Dahme, T., Krieg, L., Just, S.*, and Rottbauer, W.* (2025). Smarce1 fine-tunes cardiomyocyte proliferation in the embryonic zebrafish heart. Front Cell Dev Biol 13, 1636944. *Shared Senior Authors
Hufken T, Lobmeyer T, Gahr B, Bschorr F, Speidel T, Just S, et al. Magnetic resonance microscopy for submillimeter samples in a horizontal MR scanner. Sci Rep. 2024;14(1):23583. Epub 2024/10/10.
J. Hofeichner, B. M. Gahr, M. Huber, A. Boos, W. Rottbauer, S. Just. CRISPR/Cas9-mediated nexilin deficiency interferes with cardiac contractile function in zebrafish in vivo. Sci Rep. 2023. 13(1):22679.10.1038/s41598-023-50065-9.
L. Meier, B. M. Gahr, A. Roth, A. Gihring, S. Kirschner, C. Woitaske-Proske, J. Baier, C. Peifer, S. Just, U. Knippschild. Zebrafish as model system for the biological characterization of CK1 inhibitors. Front Pharmacol. 2023. 14:1245246.10.3389/fphar.2023.1245246.
J. Krause, A. Nickel, A. Madsen, H. M. Aitken-Buck, A. M. S. Stoter, J. Schrapers, F. Ojeda, K. Geiger, M. Kern, M. Kohlhaas, E. Bertero, P. Hofmockel, F. Hubner, I. Assum, M. Heinig, C. Muller, A. Hansen, T. Krause, D. D. Park, S. Just, D. Aissi, D. Bornigen, D. Lindner, N. Friedrich, K. Alhussini, C. Bening, R. B. Schnabel, M. Karakas, L. Iacoviello, V. Salomaa, A. Linneberg, H. Tunstall-Pedoe, K. Kuulasmaa, P. Kirchhof, S. Blankenberg, T. Christ, T. Eschenhagen, R. R. Lamberts, C. Maack, J. Stenzig, T. Zeller. An arrhythmogenic metabolite in atrial fibrillation. J Transl Med. 2023. 21(1):566.10.1186/s12967-023-04420-z.
A. Boos, B. M. Gahr, D. D. Park, V. Braun, A. Buhler, W. Rottbauer, S. Just. Hdac1-deficiency affects the cell cycle axis Cdc25-Cdk1 causing impaired G2/M phase progression and reduced cardiomyocyte proliferation in zebrafish. Biochem Biophys Res Commun. 2023. 665:98-106.10.1016/j.bbrc.2023.04.116.
S. Michels, D. Kurz, A. Rosenbohm, R. S. Peter, S. Just, H. Bazner, A. Bortlein, C. Dettmers, H. J. Gold, A. Kohler, M. Naumann, P. Ratzka, A. C. Ludolph, D. Rothenbacher, G. Nagel, J. Dorst ,A. L. S. Registry Swabia Study Group. Association of blood lipids with onset and prognosis of amyotrophic lateral sclerosis: results from the ALS Swabia registry. J Neurol. 2023. 270(6):3082-3090.10.1007/s00415-023-11630-4.
Rayhane Nchioua, Federica Diofano, Sabrina Noettger, Pascal von Maltitz, Steffen Stenger, Fabian Zech, Jan Münch, Konstantin M J Sparrer, Steffen Just, Frank Kirchhoff. Strong attenuation of SARS-CoV-2 Omicron BA.1 and increased replication of the BA.5 subvariant in human cardiomyocytes.Signal Transduct Target Ther. 2022 Dec; 7(1), 395. doi: 10.1038/s41392-022-01256-9.
Francesca Ferrante, Benedetto Daniele Giaimo, Tobias Friedrich, Toshiya Sugino, Daniel Mertens, Sabrina Kugler, Bernd Martin Gahr, Steffen Just, Leiling Pan, Marek Bartkuhn, Michael Potente, Franz Oswald, Tilman Borggrefe. Hydroxylation of the NOTCH1 intracellular domain regulates Notch signaling dynamics. Cell Death Dis. 2022 Jul 12;13(7):600. doi: 10.1038/s41419-022-05052-9.
Philipp Voisard, Federica Diofano, Amelia A Glazier, Wolfgang Rottbauer, Steffen Just. CRISPR/Cas9-Mediated Constitutive Loss of VCP (Valosin-Containing Protein) Impairs Proteostasis and Leads to Defective Striated Muscle Structure and Function In Vivo. Int J Mol Sci. 2022 Jun 16;23(12):6722. doi: 10.3390/ijms23126722.
Bühler A, Gahr B, Park DD, Bertozzi A, Boos A, Dalvoy M, Pott A, Oswald F, Kovall R, Kühn B, Weidinger G, Rottbauer W, Just S. Histone deacetylase 1 controls cardiomyocyte proliferation during embryonic heart development and cardiac regeneration in zebrafish. PLoS Genet. 2021 Nov1;17(11):e1009890.doi:10.1371/journal.pgen.1009890. eCollection 2021 Nov
Park DD, Gahr B, Krause J, Rottbauer W, Zeller T, Just S. Long-Chain Acyl-Carnitines Interfere with Mitochondrial ATP Production Leading to Cardiac Dysfunction in Zebrafish. Int J Mol Sci 2021 Aug 6;22(16):8468. doi: 10.3390/ijms22168468.
Caterina Prelli Bozzo , Rayhane Nchioua , Meta Volcic , Lennart Koepke , Jana Krüger , Desiree Schütz , Sandra Heller , Christina M Stürzel , Dorota Kmiec , Carina Conzelmann, Janis Müller , Fabian Zech , Elisabeth Braun , Rüdiger Groß , Lukas Wettstein , Tatjana Weil , Johanna Weiß , Federica Diofano , Armando A Rodríguez Alfonso, Sebastian Wiese, Daniel Sauter, Jan Münch, Christine Goffinet, Alberto Catanese, Michael Schön, Tobias M Boeckers, Steffen Stenger, Kei Sato, Steffen Just, Alexander Kleger, Konstantin M J Sparrer, Frank Kirchhoff. IFITM proteins promote SARS-CoV-2 infection and are targets for virus inhibition in vitro. Nat Commun 2021 Jul 28;12(1):4584.doi: 10.1038/s41467-021-24817-y.
Sandra Hoffmann, Ralph Roeth, Sabrina Diebold, Jasmin Gogel, David Hassel, Steffen Just, Gudrun A Rappold. Identification and Tissue-Specific Characterization of Novel SHOX-Regulated Genes in Zebrafish Highlights SOX Family Members Among Other Genes. Front Genet. 2021 May 27;12:688808. doi: 10.3389/fgene.2021.688808. eCollection 2021.
S Hoffmann, S Schmitteckert, K Raedecke, D Rheinert, S Diebold, R Roeth, B Weiss, M Granzow, B Niesler, A Griesbeck, V Eckstein, W-H Zimmermann, S Just, G A Rappold. Network-driven discovery yields new insight into Shox2-dependent cardiac rhythm control. Biochim Biophys Acta Gene Regul Mech. Apr-May 2021;1864(4-5):194702. doi: 10.1016/j.bbagrm.2021.194702. Epub 2021 Mar 8.
Daniel J Klionsky , Just S. Abdel-Aziz AK et al. Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition) Autophagy. 2021 Feb 8;1-382.doi:10.1080/15548627.2020.1797280.
Rattka M, Westphal S, Gahr B, Just S, Rottbauer W. Spen deficiency interferes with Connexin 43 expression and leads to heart failure in zebrafish. J Mol Cell Cardiol. 2021 Feb 5;155:25-35.doi: 10.1016/j.yjmcc.2021.01.006
Hao Li, Yangyang Qu, Metze P, Sommerfeld F , Just S, Abaei A, Rasche V. Quantification of Biventricular Myocardial Strain Using CMR Feature Tracking: Reproducibility in Small Animals. Biomed Res Int. 2021 Jan 22;2021:8492705. doi: 10.1155/2021/8492705
Diofano F, Weinmann K, Schneider I, Thiessen K, Rottbauer W, Just S. Genetic compensation prevents myopathy and heart failure in an in vivo model of Bag3 deficiency. PLoS Genet.2020 Nov 2;16(11):e1009088. doi: 10.1371/journal.pgen.1009088
Ferrante F; Giaimo BD; Bartkuhn M; Zimmermann T; Close V; Mertens D ; Nist A; Stiewe T; Meier-Soelch J; Kracht M; Just S; Kloble P; Oswald F; Borggrefe T. HDAC3 functions as a positive regulator in Notch signal transduction. Nucleic Acids Res. 2020 Apr 17;48(7):3496-3512. doi: 10.1093/nar/gkaa088.
Li H; Metze P; Abaei A; Rottbauer W; Just S; Lu QH ; Rasche V. Feasibility of real-time cardiac MRI in mice using tiny golden angle radial sparse. NMR Biomed. 2020 Jul;33(7):e4300. doi: 10.1002/nbm.4300
Li H; Abaei A; Metze P; Just S; Lu QH; Rasche V. Technical Aspects ofin vivoSmall Animal CMR Imaging. FRONTIERS IN PHYSICS, Review, JUN 10 2020, DOI: 10.3389/fphy.2020.00183
Pott A, Rottbauer W, Just S. Streamlining drug discovery assays for cardiovascular disease using zebrafish. Expert Opinion in Drug Discovery. 2019 Oct 01. doi.org/10.1080/17460441.2020.1671351
Schutera M, Just S, Gierten J, Mikut R, Reischl M, Pylatiuk C. Machine Learning Methods for Automated Quantification of Ventricular Dimensions. Zebrafish. 2019 Sep 19. doi: 10.1089/zeb.2019.1754.
Hoffmann S, Paone C, Sumer SA, Diebold S, Weiss B, Roeth R, Clauss S, Klier I, Kääb S, Schulz A, Wild PS, Ghrib A, Zeller T, Schnabel RB, Just S, Rappold GA. Functional Characterization of Rare Variants in the SHOX2 Gene Identified in Sinus Node Dysfunction and Atrial Fibrillation. Front Genet. 2019 Jul 11;10:648. doi: 10.3389/fgene.2019.00648
Lausser L*, Siegle L*, Rottbauer W, Frank D**, Just S**, Kestler HA**. Semantic Multi-Classifier Systems Identify Predictive Processes in Heart Failure Models across Species. *equal contribution, **joint senior authors, Biomolecules 2018, 8(4), 158; doi.org/10.3390/biom8040158
Pott A, Just S. Metabolic Profiling of Glucocorticoid Deficiency: A "Fishing" Expedition. EBioMedicine. 2018 Oct 31. pii: S2352-3964(18)30476-6. doi: 10.1016/j.ebiom.2018.10.058.
Pott A*, Shahid M*, Köhler D, Pylatiuk C, Weinmann K, Just S** and Rottbauer W** Therapeutic Chemical Screen Identifies Phosphatase Inhibitors to Reconstitute PKB Phosphorylation and Cardiac Contractility in ILK-Deficient Zebrafish. *equal contribution, **joint senior authors, Biomolecules 2018, 8(4), 153; doi:10.3390/biom8040153
Krämer LM, Brettschneider J, Lennerz JK, Walcher D, Fang L, Rosenbohm A, Balakrishnan K, Benckendorff J, Möller P, Just S, Willem M, Ludolph AC, Thal DR. Amyloid precursor protein-fragments-containing inclusions in cardiomyocytes with basophilic degeneration and its association with cerebral amyloid angiopathy and myocardial fibrosis.Sci Rep. 2018 Nov 9;8(1):16594. doi: 10.1038/s41598-018-34808-7.
Segert J, Schneider I, Berger IM, Rottbauer W, Just S. Mediator complex subunit Med12 regulates cardiac jelly development and AV valve formation in zebrafish. Prog Biophys Mol Biol. 2018 Jul 20. pii: S0079-6107(18)30023-3. doi: 10.1016/j.pbiomolbio.2018.07.010.
Kustermann M, Manta L, Paone C, Kustermann J, Lausser L, Wiesner C, Eichinger L, Clemen CS, Schröder R, Kestler HA, Sandri M, Rottbauer W, Just S. Loss of the novel Vcp (valosin containing protein) interactor Washc4 interferes with autophagy-mediated proteostasis in striated muscle and leads to myopathy in vivo. Autophagy. 2018;14(11):1911-1927. doi: 10.1080/15548627.2018.1491491. Epub 2018 Aug 16.
Helferich AM, Brockmann SJ, Reinders J, Deshpande D, Holzmann K, Brenner D, Andersen PM, Petri S, Thal DR, Michaelis J, Otto M, Just S, Ludolph AC, Danzer KM, Freischmidt A, Weishaupt JH. Dysregulation of a novel miR-1825/TBCB/TUBA4A pathway in sporadic and familial ALS. Cell Mol Life Sci. 2018 Dec;75(23):4301-4319. doi: 10.1007/s00018-018-2873-1. Epub 2018 Jul 20.
Kramer F, Just S, Zeller T. New Perspectives: Systems medicine in cardiovascular disease. BMC Systems Biology ; 2018 Apr 25. doi: 10.1186/s12918-018-0579-5
Paone C, Diofano F, Park DD, Rottbauer W, Just S. Genetics of Cardiovascular Disease: Fishing for Causality. Frontiers in Cardiovascular Medicine. Front. Cardiovasc. Med., 01 June 2018, doi.org/10.3389/fcvm.2018.00060
Pott A, Bock S, Berger IM, Frese K, Dahme T, Keßler M, Rinné S, Decher N, Just S*, Rottbauer W*. Mutation of the Na+/K+-ATPase Atp1a1a.1 causes QT interval prolongation and bradycardia in zebrafish. * equal contribution. J Mol Cell Cardiol. 2018 Jul;120:42-52. doi: 10.1016/j.yjmcc.2018.05.005. Epub 2018 May 8.
Keßler M, Kieltsch A, Kayvanpour E, Katus HA, Schoser B, Schessl J, Just S*, Rottbauer W* (2018) A zebrafish model for FHL1-opathy reveals loss-of-function effects of human FHL1 mutations. * equal contribution. Neuromuscular Disorders doi:10.1016/j.nmd.2018.03.001
Silbernagel N, Walecki M, Schäfer MK, Kessler M, Zobeiri M, Rinné S, Kiper AK, Komadowski MA, Vowinkel KS, Wemhöner K, Fortmüller L, Schewe M, Dolga AM, Scekic-Zahirovic J, Matschke LA, Culmsee C, Baukrowitz T, Monassier L, Ullrich ND, Dupuis L, Just S, Budde T, Fabritz L & Decher N. The VAMP-associated protein VAPB is required for cardiac and neuronal pacemaker channel function. FASEB Journal. In press doi.org/10.1096/fj.201800246R
Brockmann SJ, Freischmidt A, Oeckl P, Muller K, Ponna SK, Helferich AM, Paone C, Reinders J, Kojer K, Orth M, Jokela M, Auranen M, Udd B, Hermann A, Danzer KM, Lichtner P, Walther P, Ludolph AC, Andersen PM, Otto M, Kursula P, Just S, Weishaupt JH (2018) CHCHD10 mutations p.R15L and p.G66V cause motoneuron disease by haploinsufficiency. Hum Mol Genet doi:10.1093/hmg/ddx436
Paone C, Rudeck S, Etard C, Strahle U, Rottbauer W, Just S (2018) Loss of zebrafish Smyd1a interferes with myofibrillar integrity without triggering the misfolded myosin response. Biochem Biophys Res Commun doi:10.1016/j.bbrc.2018.01.060
Decher N, Ortiz-Bonnin B, Friedrich C, Schewe M, Kiper AK, Rinné S, Seemann G, Peyronnet R, Zumhagen S, Bustos D, Kockskämper J, Kohl P, Just S, González W, Baukrowitz T, Stallmeyer B, Schulze-Bahr E. Sodium permeable and "hypersensitive" TREK-1 channels cause ventricular tachycardia.EMBO Mol Med. 2017 Feb 27, doi: 10.15252/emmm.201606690
Straubinger J, Boldt K, Kuret A, Deng L, Krattenmacher D, Bork N, Desch M, Feil R, Feil S, Nemer M, Ueffing M, Ruth P, Just S, Lukowski R. Amplified pathogenic actions of angiotensin II in cysteine-rich LIM-only protein 4 negative mouse hearts. FASEB Journal. 2017 Jan 30, doi: 10.1096/fj.201601186
Just S, Raphel L, Berger IM, Bühler A, Keßler M, Rottbauer W. Tbx20 Is an Essential Regulator of Embryonic Heart Growth in Zebrafish.2016 Dec 1, doi: 10.1371/journal.pone.0167306. eCollection 2016
Just S, Hirth S, Berger IM, Fishman MC, Rottbauer W. The mediator complex subunit Med10 regulates heart valve formation in zebrafish by controlling Tbx2b-mediated Has2 expression and cardiac jelly formation.Biochem Biophys Res Commun. 2016 Jun 22, doi:10.1016/j.bbrc.2016.06.088
Rudeck S, Etard C, Khan MM, Rottbauer W, Rudolf R, Strähle U, Just S. A compact unc45b-promoter drives muscle-specific expression in zebrafish and mouse.Genesis. 2016 Jun 13, doi: 10.1002/dvg.22953
Richter J, Rudeck S, Kretz AL, Kramer K, Just S, Henne-Bruns D, Hillenbrand A, Leithäuser F, Lemke J, Knippschild U. Decreased CK1δ expression predicts prolonged survival in colorectal cancer patients.Tumour Biol. 2016 Jan 7, doi: 10.1007/s13277-015-4745-8
Hirth S, Bühler A, Bührdel JB, Rudeck S, Dahme T, Rottbauer W, Just S. Paxillin and Focal Adhesion Kinase (FAK) Regulate Cardiac Contractility in the Zebrafish Heart.PLoS One. 2016 Mar 8, doi: 10.1371/journal.pone.0150323. eCollection 2016
Hoffmann S, Clauss S, Berger IM, Weiß B, Montalbano A, Röth R, Bucher M, Klier I, Wakili R, Seitz H, Schulze-Bahr E, Katus HA, Flachsbart F, Nebel A, Guenther SP, Bagaev E, Rottbauer W, Kääb S, Just S, Rappold GA. Coding and non-coding variants in the SHOX2 gene in patients with early-onset atrial fibrillation.Basic Res Cardiol. 2016 May, doi: 10.1007/s00395-016-0557-2
Bühler A, Kustermann M, Bummer T, Rottbauer W, Sandri M, Just S. Atrogin-1 Deficiency Leads to Myopathy and Heart Failure in Zebrafish.Int J Mol Sci. 2016 Jan 30, doi: 10.3390/ijms17020187
Aherrahrou Z, Schlossarek S, Stoelting S, Klinger M, Geertz B, Weinberger F, Kessler T, Aherrahrou R, Just S, Rottbauer R, Eschenhagen T, Schunkert H, Carrier L, Erdmann J. Knock-out of nexilin in mice leads to dilated cardiomyopathy and endomyocardial fibroelastosis. Basic Res Cardiol. 2016 Jan, doi: 10.1007/s00395-015-0522-5
Kessler M, Rottbauer W, Just S. Recent progress in the use of zebrafish for novel cardiac drug discovery.Expert opinion on drug discovery. 2015 Nov 2, doi: 10.1517/17460441.2015.1078788
Frese KS, Meder B, Keller A, Just S, Haas J, Vogel B, Fischer S, Backes C, Matzas M, Kohler D, Benes V, Katus HA, Rottbauer W. RNA splicing regulated by RBFOX1 is essential for cardiac function in zebrafish.Journal of cell science. 2015 Aug 15, doi: 10.1242/jcs.166850
Clemen CS, Marko M, Strucksberg KH, Behrens J, Wittig I, Gartner L, Winter L, Chevessier F, Matthias J, Turk M, Tangavelou K, Schutz J, Arhzaouy K, Klopffleisch K, Hanisch FG, Rottbauer W, Blumcke I, Just S, Eichinger L, Hofmann A, Schroder R. VCP and PSMF1: Antagonistic regulators of proteasome activity. Biochem Biophys Res Commun. 2015 Aug 7, doi: 10.1016/j.bbrc.2015.06.086
Zebrowski DC, Vergarajauregui S, Wu CC, Piatkowski T, Becker R, Leone M, Hirth S, Ricciardi F, Falk N, Giessl A, Just S, Braun T, Weidinger G, Engel FB. Developmental alterations in centrosome integrity contribute to the post-mitotic state of mammalian cardiomyocytes.eLife. 2015 Aug 6, doi: 10.7554/eLife.05563
Buhrdel JB, Hirth S, Kessler M, Westphal S, Forster M, Manta L, Wiche G, Schoser B, Schessl J, Schroder R, Clemen CS, Eichinger L, Furst DO, van der Ven PF, Rottbauer W, Just S. In vivo characterization of human myofibrillar myopathy genes in zebrafish. Biochem Biophys Res Commun. 2015 May 29, doi: 10.1016/j.bbrc.2015.03.149
Hein K, Mittler G, Cizelsky W, Kuhl M, Ferrante F, Liefke R, Berger IM, Just S, Strang JE, Kestler HA, Oswald F and Borggrefe T. Site-specific methylation of Notch1 controls the amplitude and duration of the Notch1 response.Science signaling. 2015 Mar 24, doi: 10.1126/scisignal.2005892
Kessler M, Berger IM, Just S* and Rottbauer W*. Loss of dihydrolipoyl succinyltransferase (DLST) leads to reduced resting heart rate in the zebrafish. BasicRes Cardiol. 2015 Mar, doi: 10.1007/s00395-015-0468-7, * shared senior authorship
Clemen CS, Stockigt F, Strucksberg KH, Chevessier F, Winter L, Schutz J, Bauer R, Thorweihe JM, Wenzel D, Schlotzer-Schrehardt U, Rasche V, Krsmanovic P, Katus HA, Rottbauer W, Just S, Muller OJ, Friedrich O, Meyer R, Herrmann H, Schrickel JW, Schroder R. The toxic effect of R350P mutant desmin in striated muscle of man and mouse.Acta Neuropathol. 2015 Feb, doi: 10.1007/s00401-014-1363-2
Pott A, Rottbauer W, Just S. Functional genomics in zebrafish as a tool to identify novel antiarrhythmic targets. Current medicinal chemistry. 2014 Apr, 2014;21(11):1320-9. Review
Spomer W, Pfriem A, Alshut R, Just S, Pylatiuk C. High-throughput screening of zebrafish embryos using automated heart detection and imaging.Journal of laboratory automation. 2012 Dec, doi: 10.1177/2211068212464223
Just S, Berger IM, Meder B, Backs J, Keller A, Marquart S, Frese K, Patzel E, Rauch GJ, Katus HA, Rottbauer W. Protein kinase d2 controls cardiac valve formation in zebrafish by regulating histone deacetylase 5 activity.Circulation. 2011 Jul 19, doi: 10.1161/CIRCULATIONAHA.110.003301
Just S, Meder B, Berger IM, Etard C, Trano N, Patzel E, Hassel D, Marquart S, Dahme T, Vogel B, Fishman MC, Katus HA, Strahle U, Rottbauer W. The myosin-interacting protein smyd1 is essential for sarcomere organization.J Cell Sci. 2011 Oct 1, doi: 10.1242/jcs.084772
Hassel D* , Dahme T* , Erdmann J* , Meder B, Huge A, Stoll M, Just S, Hess A, Ehlermann P, Weichenhan D, Grimmler M, Liptau H, Hetzer R, Regitz-Zagrosek V, Fischer C, Nurnberg P, Schunkert H, Katus HA, Rottbauer W. Nexilin mutations destabilize cardiac z-disks and lead to dilated cardiomyopathy. Nat Med. 2009 Nov, doi: 10.1038/nm.2037 * Erstautoren
Thum T, Gross C, Fiedler J, Fischer T, Kissler S, Bussen M, Galuppo P, Just S, Rottbauer W, Frantz S, Castoldi M, Soutschek J, Koteliansky V, Rosenwald A, Basson MA, Licht JD, Pena JT, Rouhanifard SH, Muckenthaler MU, Tuschl T, Martin GR, Bauersachs J, Engelhardt S. MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts.Nature. 2008 Dec 18, doi: 10.1038/nature07511
Rottbauer W*, Just S*, Wessels G, Trano N, Most P, Katus HA, Fishman MC. VEGF-PLC gamma1 pathway controls cardiac contractility in the embryonic heart.Genes Dev. 2005 Jul 1, doi: 10.1101/gad.1319405, * Erstautoren
Job Offers
We offer up to two positions each semester.
Our MD students are expected to apply for the program "Experimental Medicine".
If you’re interested feel free to contact Mirjam Glöckler.
We are always looking for interested and motivated students. Feel free to contact Mirjam Glöckler or one of our PostDocs.
Various options for internships are possible. Feel free to contact Mirjam Glöckler or one of our PostDocs.