Head

Prof. Miriam Erlacher, MD PhD
Ulm University Medical Center
Department of Pediatrics and Adolescent Medicine
Eythstr. 24
89075 Ulm, Germany
phone: +49-731-500 57000
e-mail: direktion.kinder@uniklinik-ulm.de

Dr. Juncal Fernandez-Orth, Junior research leader
Dr. Jovana Rajak, PhD
Marina Alcaide, Phd-Stud.
Helgard Knauss, CTA
Daniel Mader, CS
Shuhan Yin, Phd-Stud.
Research Themes
Our research focuses on the role of apoptosis regulated by BCL-2 proteins in the healthy and the diseased hematopoietic system, with special focus on human hematopoiesis, rare leukemias of childhood (e.g. juvenile myelomonocytic leukemia, MDS) and inherited bone marrow failure syndromes (e.g. dyskeratosis congenita, Fanconi anemia). We also aim at understanding syndromes predisposing to leukemia and the specific mechanisms of malignant transformation. Finally, it is our goal to generate model systems reminiscent of human disease and suitable for research with high translational potential.
Apoptosis in the hematopoietic system and during leukemogenesis
As in other tissues, a tight regulation of survival and death decisions is required for maintenance, homeostasis and function of the hematopoietic system. Deregulation of apoptosis signaling contributes to many hematological diseases characterized by cytopenia (e.g. severe congenital neutropenia, bone marrow failure syndromes etc.). In addition, apoptosis signaling and its deregulation play major roles during leukemogenesis, and apoptosis resistance is regarded as one of the “hallmarks of cancer” required for full malignant transformation. We extensively studied deregulation of apoptosis signailng as driver of malignant transformation in juvenile myelomonocytic leukemia (Wu et al, Leukemia, 2024; Koleci et al, Cell Death Dis, 2025).
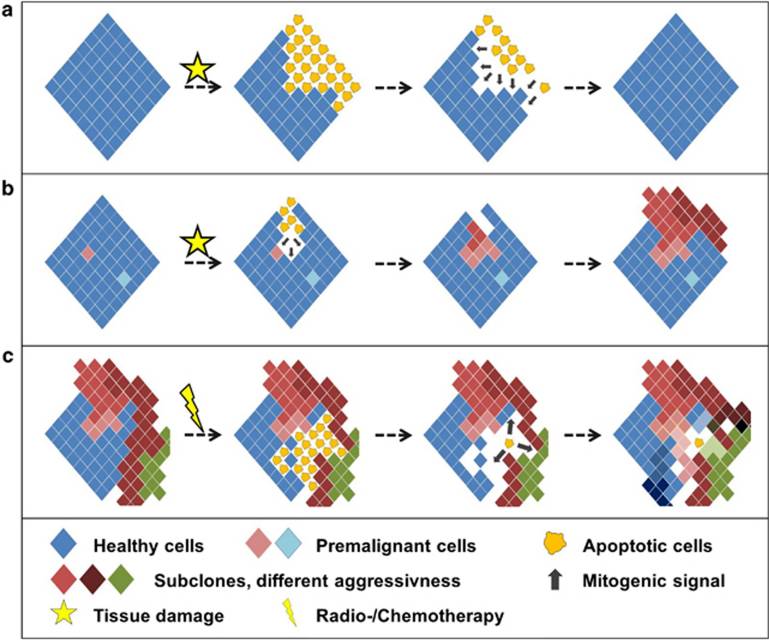
There is, however, emerging evidence that apoptosis of premalignant cells does not always act as a barrier against malignant transformation but rather can drive compensatory proliferation and selection of individual clones. This drives, on a long run, genomic instability and facilitates the emergence of malignant clones (Figure 1). In a mouse model of dyskeratosis congenita, we indeed could show that apoptosis drives both bone marrow failure and leukemia: Genetic inhibition of PUMA-induced apoptosis was sufficient to maintain hematopoeisis and genomic stability in this model system for bone marrow failure (Molnar et al, Cell Death Diff, 2025).
Current Projects
CRC 1479: Oncogene-driven immune escape (OncoEscape)
Speaker: Prof. Dr. R. Zeiser, Medical Center University of Freiburg - Project Ulm by Prof. M. Erlacher
CRC/Transregio 353: Regulation of Cell Death Decisions
Speaker: Prof. Dr. Th. Brunner, University of Konstanz - Project Ulm by Prof. M. Erlacher
MyPred - Predisposition to Myeloid Neoplasms
Speaker: Prof. Dr. M. Erlacher, Department of Pediatrics and Adolescent Medicine
How GATA2 Deficiency affects development and disease (3D-GATA2)
Speaker: Prof. M. Pasquet, Toulouse Cancer Research Center - Project Ulm by Prof. M. Erlacher
Publications
- Molnar C, Rajak J, Weiss JM, Gonzalez-Menendez I, Andrieux G, Schreiber F, Kornemann EM, Wendeburg L, Göhring G, Strahm B, Beier F, Steinemann D, Börries M, Rudelius M, Quintanilla-Martinez L, Niemeyer CM, Niewisch MR, Labi V, Bohler S, Erlacher M. PUMA-induced apoptosis drives bone marrow failure and genomic instability in telomerase-deficient mice. Cell Death Differ. 2025 Aug 19. doi: 10.1038/s41418-025-01557-w. Online ahead of print. PMID: 40830245
- Fernandez-Orth J, Koyunlar C, Weiss JM, Gioacchino E, de Looper H, Andrieux G, Ter Borg M, Zink J, Gonzalez-Menendez I, Hoogenboezem R, Yigit B, Gussinklo KJ, Mulet-Lazaro R, Wantzen C, Pfeiffer S, Molnar C, Bindels E, Bohler S, Sanders M, Quintanilla-Martinez L, Wlodarski M, Boerries M, Touw IP, Niemeyer C, Erlacher M*, de Pater E*. Hematological phenotypes in GATA2 deficiency syndrome arise from aging, maladaptation to proliferation, and somatic events. Blood Adv. 2025 Jun 10;9(11):2794-2807. doi: 10.1182/bloodadvances.2024015106. PMID: 40138552; PMCID: PMC12167812 *Authors contributed equally to this work
- Koleci N, Wu Y, Wehner NA, Rajak J, Mittapalli VR, Mergner J, Xiao H, Wang J, Wahl M, Bohler S, Aumann K, Häcker G, Ramamoorthy S, Boerries M, Kirschnek S, Erlacher M. Oncogenic and microenvironmental signals drive cell type specific apoptosis resistance in juvenile myelomonocytic leukemia. Cell Death Dis. 2025 Mar 8;16(1):165. doi: 10.1038/s41419-025-07479-2. PMID: 40057493; PMCID: PMC11890777
- Schwarz-Furlan S, Gengler C, Yoshimi-Noellke A, Piontek G, Schneider-Kimoto Y, Schmugge M, Thiede C, Niemeyer CM, Erlacher M, Rudelius M. Diagnostic features in paediatric MDS-EB with UBTF-internal tandem duplication: defining a unique subgroup. Histopathology. 2025 Mar;86(4):603-610. doi: 10.1111/his.15378. Epub 2024 Nov 20. PMID: 39564724; PMCID: PMC11791721
- Wu Y, Zehnle PMA, Rajak J, Koleci N, Andrieux G, Gallego-Villar L, Aumann K, Boerries M, Niemeyer CM, Flotho C, Bohler S, Erlacher M. BH3 mimetics and azacitidine show synergistic effects on juvenile myelomonocytic leukemia. Leukemia. 2024 Jan;38(1):136-148. doi: 10.1038/s41375-023-02079-5. Epub 2023 Nov 9. PMID: 37945692; PMCID: PMC10776398
- Erlacher M, Andresen F, Sukova M, Stary J, De Moerloose B, Bosch JVWT, Dworzak M, Seidel MG, Polychronopoulou S, Beier R, Kratz CP, Nathrath M, Frühwald MC, Göhring G, Bergmann AK, Mayerhofer C, Lebrecht D, Ramamoorthy S, Yoshimi A, Strahm B, Wlodarski MW, Niemeyer CM. Spontaneous remission and loss of monosomy 7: a window of opportunity for young children with SAMD9L syndrome. Haematologica. 2024 Feb 1;109(2):422-430. doi: 10.3324/haematol.2023.283591. PMID: 37584291; PMCID: PMC10828767
- Sahoo SS, Pastor VB, Goodings C, Voss RK, Kozyra EJ, Szvetnik A, Noellke P, Dworzak M, Starý J, Locatelli F, Masetti R, Schmugge M, De Moerloose B, Catala A, Kállay K, Turkiewicz D, Hasle H, Buechner J, Jahnukainen K, Ussowicz M, Polychronopoulou S, Smith OP, Fabri O, Barzilai S, de Haas V, Baumann I, Schwarz-Furlan S; European Working Group of MDS in Children (EWOG-MDS); Niewisch MR, Sauer MG, Burkhardt B, Lang P, Bader P, Beier R, Müller I, Albert MH, Meisel R, Schulz A, Cario G, Panda PK, Wehrle J, Hirabayashi S, Derecka M, Durruthy-Durruthy R, Göhring G, Yoshimi-Noellke A, Ku M, Lebrecht D, Erlacher M, Flotho C, Strahm B, Niemeyer CM, Wlodarski MW. Publisher Correction: Clinical evolution, genetic landscape and trajectories of clonal hematopoiesis in SAMD9/SAMD9L syndromes. Nat Med. 2021 Dec;27(12):2248. doi: 10.1038/s41591-021-01632-y. Erratum for: Nat Med. 2021 Oct;27(10):1806-1817. doi: 10.1038/s41591-021-01511-6. PMID: 34799732
- Bohler S, Afreen S, Fernandez-Orth J, Demmerath EM, Molnar C, Wu Y, Weiss JM, Mittapalli VR, Konstantinidis L, Schmal H, Kunze M, Erlacher M. Inhibition of the anti-apoptotic protein MCL-1 severely suppresses human hematopoiesis. Haematologica. 2021 Dec 1;106(12):3136-3148. doi: 10.3324/haematol.2020.252130. PMID: 33241675; PMCID: PMC8634190
- Krombholz CF, Gallego-Villar L, Sahoo SS, Panda PK, Wlodarski MW, Aumann K, Hartmann M, Lipka DB, Daskalakis M, Plass C, Niemeyer CM, Erlacher M*, Flotho C*. Azacitidine is effective for targeting leukemia-initiating cells in juvenile myelomonocytic leukemia. Leukemia. 2019 Jul;33(7):1805-1810. doi: 10.1038/s41375-018-0343-2. Epub 2019 Jan 24. PMID: 30679798 *Authors contributed equally to this work
- Kollek M, Voigt G, Molnar C, Murad F, Bertele D, Krombholz CF, Bohler S, Labi V, Schiller S, Kunze M, Geley S, Niemeyer CM, Garcia-Saez A, Erlacher M. Transient apoptosis inhibition in donor stem cells improves hematopoietic stem cell transplantation. J Exp Med. 2017 Oct 2;214(10):2967-2983. doi: 10.1084/jem.20161721. Epub 2017 Sep 7. PMID: 28882984; PMCID: PMC5626392.