Head

Prof. Dr. rer. nat. Gudrun Strauß
Ulm University Medical Center
Department of Pediatrics and Adolescent Medicine
Eythstr. 24 (Research Lab, House 16)
89075 Ulm, Germany
phone: +49-731-500 57033
e-mail: Gudrun.Strauss@uniklinik-ulm.de

name | position | |
Strauß, Gudrun | Head | gudrun.strauss@uniklinik-ulm.de |
Kawach, Rawan Dr. | Post-Doc | rawan.kawach@uni-ulm.de |
Wahl, Lena | Phd student | lena.wahl@uni-ulm.de |
Knape, Ingrid | Technician | ingrid.knape@uniklinik-ulm.de |
Alumnis 2020 - 2025 |
| |
Dasari, Prasad Dr. | Post-Doc |
|
Enders, Matthias Dr. | Post-Doc |
|
Scheurer, Jasmin | Phd student |
|
Reiser, Tanja | Phd student |
|
Liu, Jianing | MD student |
|
Saleh, Kara | MD student |
|
Greiner, Jennifer | MD student |
|
Keltsch, Emma | MD student |
|
Meltzner, Annika | Master student |
|
Diamante, Gabriele | Master student |
|
Weber, Sophie | Bacheolor student |
|
Rezzaei, Melika | Technican |
|
We welcome applications for Master's theses and medical doctoral theses.
Research Profile
In our research group we investigate the regulation of the immune response to various insults ranging from tumor development, trauma and sepsis up to aging processes. Our main interest concerns the multiple functions of myeloid-derived suppressor cells (MDSCs) to shape innate and adaptive immunity according to different pathophysiological stimuli.
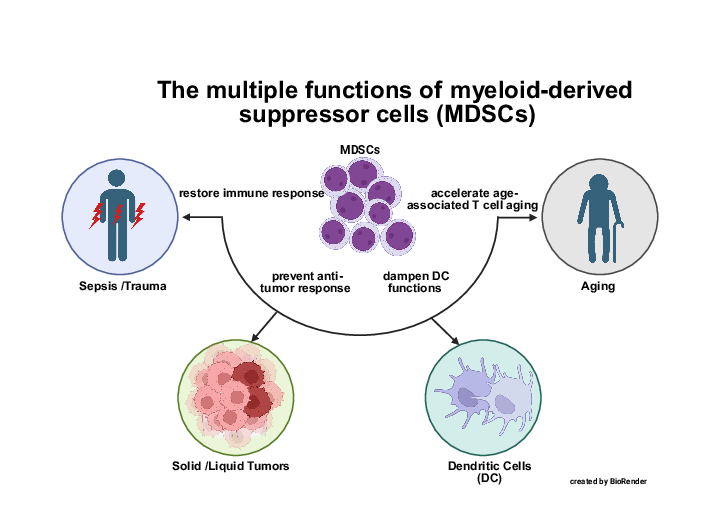
MDSCs: a therapeutic option to counterbalance sepsis-induced immune paralysis
Sepsis is one of the most leading courses in death worldwide. While early deaths induced by hyperinflammation and organ failure are constantly decreasing due to improved ICU treatment, late death attributed by severe immunosuppression and secondary infections are currently difficult to treat. In our murine sepsis model and in in vitro models of human sepsis, MDSCs prevent sepsis induced death and restore immune functions. Current research focus on the impact on MDSCs on reprogramming of T cells, innate immunity and microbiome.
MDSCs: modulators of immune responses in liquid and solid tumors
Tumor development is associated with expansion of intratumoral and peripheral MDSCs. In human ovarian cancer patients and childhood acute lymphoblastic leukemias, we are analyzing the impact of MDSCs on the underlying anti-tumor response. Current research focus on defining molecular tumor subtypes associated with MDSC induction and identifying tumor-derived MDSC-inducing factors.
MDSCs: Contributors to immunosenescence
Immune cells are strongly affected by aging, a process named immunosenescence, which is responsible for inefficient immune responses leading to increased susceptibility for infections, tumor development or inefficient vaccination responses. By comparing young and old healthy individuals, we could show that MDSC are increased and change their function in old individuals and contribute to age-associated impaired T cell responses.
1. Keltsch, E., Greiner, J., Wahl, L., Knape, I., Tews, D., Denkinger, M., Debatin, K. M., and Strauss, G. (2025) Aging modulates the immunosuppressive, polarizing and metabolic functions of blood-derived myeloid-derived suppressor cells (MDSCs). Immun Ageing22, 29
2. Mosenlechner, M., Schlosser, D., Braumuller, S., Dorfer, L., Mannes, M., Kawach, R., Strauss, G., Schmidt, C. Q., Lupu, L., and Huber-Lang, M. S. (2025) Induction of Early Pulmonary Senescence in Experimental Sepsis. Shock63, 448-455
3. Tschaffon-Muller, M. E. A., Kempter, E., Steppe, L., Kupfer, S., Kuhn, M. R., Gebhard, F., Pankratz, C., Kalbitz, M., Schutze, K., Gundel, H., Kaleck, N., Strauss, G., Vacher, J., Ichinose, H., Weimer, K., Ignatius, A., Haffner-Luntzer, M., and Reber, S. O. (2023) Neutrophil-derived catecholamines mediate negative stress effects on bone. Nat Commun14, 3262
4. Kustermann, M., Dasari, P., Knape, I., Keltsch, E., Liu, J., Pfluger, S., Osen, W., Holzmann, K., Huber-Lang, M., Debatin, K. M., and Strauss, G. (2023) Adoptively Transferred in vitro-Generated Myeloid-Derived Suppressor Cells Improve T-Cell Function and Antigen-Specific Immunity after Traumatic Lung Injury. J Innate Immun15, 78-95
5. Kempter, E., Amoroso, M., Kupfer, S., Lupu, L., Kustermann, M., Scheurer, J., Baumann, B., Wirth, T., Gundel, H., Straub, R. H., Strauss, G., Huber-Lang, M., Langgartner, D., and Reber, S. O. (2023) The PMN-MDSC - A key player in glucocorticoid resistance following combined physical and psychosocial trauma. Brain Behav Immun108, 148-161
6. Scheurer, J., Kitt, K., Huber, H. J., Fundel-Clemens, K., Pflanz, S., Debatin, K. M., and Strauss, G. (2021) Graft-Versus-Host Disease Prevention by In Vitro-Generated Myeloid-Derived Suppressor Cells Is Exclusively Mediated by the CD11b+CD11c+ MDSC Subpopulation. Front Immunol12, 754316
7. Reisser, T., Halbgebauer, D., Scheurer, J., Wolf, L., Leithauser, F., Beyersdorf, N., Fischer-Posovszky, P., Debatin, K. M., and Strauss, G. (2020) In vitro-generated alloantigen-specific Th9 cells mediate antileukemia cytotoxicity in the absence of graft-versus-host disease. Leukemia34, 1943-1948
8. Kustermann, M., Klingspor, M., Huber-Lang, M., Debatin, K. M., and Strauss, G. (2019) Immunostimulatory functions of adoptively transferred MDSCs in experimental blunt chest trauma. Sci Rep9, 7992
