Chronic stress is a burden of modern societies and is known to be a risk factor for numerous somatic and affective disorders, including cardiovascular dysfunction, inflammatory bowel disease, gastric ulceration, general and/ or social anxiety- and depression-related diseases, as well as systemic immunological dysfunctions. Although many of these disorders are paralleled by either hypocorticism, glucocorticoid (GC) resistance, or a combination of both, resulting in a decreased GC signalling, the underlying aetiology of these disorders is still poorly understood, at least partly due to a lack of appropriate and clinically relevant animal models.
Importantly, not all people are equally affected by chronic stressors. While some individuals subjected to severe adverse life events develop pathologies, others do not. But again, the underlying mechanisms engendering resilience and vulnerability are also far from being understood and, thus, the possibility to clinically benefit from these observations is currently still limited.
A failure of immunoregulation, attributable to reduced exposure to the microbial environment within which the mammalian immune system evolved ("Old friends hypothesis"), is thought to be one factor contributing to recent increases in stress-related chronic inflammatory disorders, as well as mood disorders for which chronic low-grade inflammation is a risk factor ("cytokine theory of depression") (Lowry et al., 2016 Curr Environ Health Rep; Reber et al., 2016 PNEC).
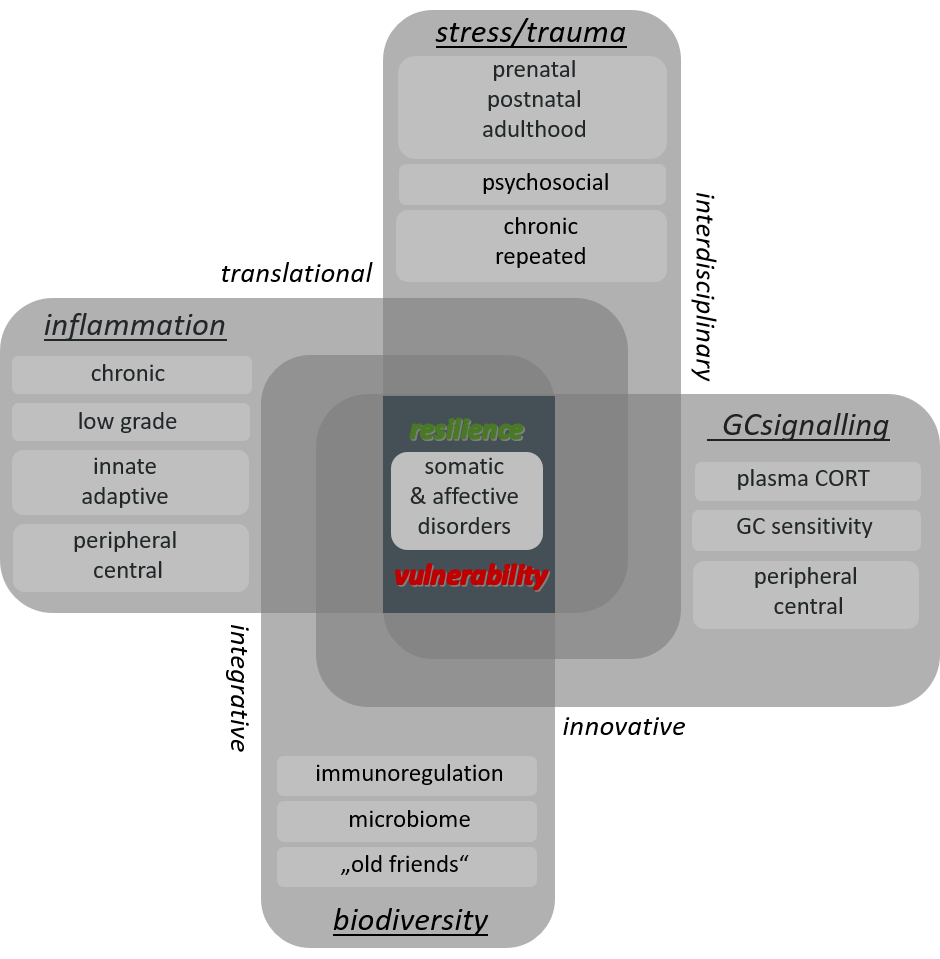
Therefore, the main research aims of the Laboratory for Molecular Psychosomatics are to extend the current knowledge on the mechanisms underlying 1i) stressor-induced development of somatic and mental pathologies and 1ii) individual differences in stress resilience and 2) to use this mechanistic knowledge for the development of novel strategies in terms of stress protection (Fig. 1).
Funding Identity of the Laboratory for Molecular Psychosomatics

Mechanisms underlying the intergenerational effects of Mycobacterium vaccae ATCC 15483 to promote stress resilience in mice. (German Research Foundation, RE 2911/23-3; sucessfully applied for by S. O. Reber).

Effects of Lactobacillus reuteri DSM 17938 on the acute psychological, endocrine and immunological stress response in healthy city dwellers. (German Research Foundation, RE 2911/26-1; successfully applied for by S.O. Reber).
This partnership aims to build research capacities, foster long-term collaboration, and lay the groundwork for raising future international funding for exploring how brain-body integration, circadian rhythms, and environmental stress influence immune function. (successfully applied for by Prof. Dr. Ilia Karatsoreos (Department of Psychological and Brain Sciences at University of Massachusetts Amherst, USA) and Prof. Dr. Stefan Reber at Ulm University Medical Center).

The role of circulating small extracellular vesicles in chronic psychosocial stress/ PTSD- related inflammation. (German Research Foundation, LA 5673/3-1; successfully applied for by D. Langgartner).

Investigating the role of circulating small extracellular vesicles in chronic psychosocial stress-/ PTSD-related inflammation. (Collaborative Research Consortium (CRC) 1149/ German Research Foundation; successfully applied for by Dr. D. Langgartner)

Effects of early life stress on bone homeostasis and fracture healting. (German Research Foundation, Project B06 within the CRC 1149, INST 40/599-1; successfully applied for by S.O. Reber and M. Haffner-Luntzer (Co-PI, Institute of Ofthopaedic Research and Biomechanics, Ulm University)).

Mycobacterium vaccae immunization: Inducing resilience to stress during pregnany in the dam and protecting her offspring. (German Research Foundation, RE 2911/23-1; successfully applied for by S.O. Reber Ulm and D.A. Slattery (Co-PI, SL141/6-1, Laboratory for Translational Psychiatry, Department of Psychiatry, Psychosomatics and Psychotherapy, Goethe University Frankfurt))

Facilitating stress resilience by "Old friends": Immunization with Mycobacterium vaccae prevents the negative effects of early life stress on chronic stress vulnerability during adulthood. (German Research Foundation, RE 2911/21-1; successfully applied for by S.O. Reber)
Stress-protective effects of Mycolicibacterium aurum Aogashima. (successfully applied for by S.O. Reber)

Facilitating resilience to repeated hits of psychosocial traumatization by "Old friend" organisms – a closer look at the influence of the intestinal microbiome. (Collaborative Research Consortium (CRC) 1149/ German Research Foundation; successfully applied for by D. Langgartner)

Effects of psychosocial trauma on bone homeostasis and fracture healing. (German Research Foundation, Project B06 within the CRC 1149, INST 40/599-1; successfully applied for by S.O. Reber Ulm and M. Haffner-Luntzer (Co-PI, Institute of Orthopaedic Research and Biomechanics, Ulm University))

Ph.D. stipend for Sandra Förtsch from April to December 2018. (International Graduate School in Molecular Medicine of Ulm University; successfully applied for by S.O. Reber)

Promotion of stress resilience by non-invasive immunoregulatory approaches.' (Office of Naval Research Global, N00014-17-S-B001; Grant12274897; successfully applied for by S.O.Reber)

Additive effects of multiple psychosocial traumatic life experiences on subsequent blunt thorax trauma. ("Bausteinprogramm" of the medical faculty of the Ulm University; successfully applied for by D. Langgartner)

Danger response and regeneration following musculoskeletal trauma: interdisciplinary approach to link physical and psychological injuries. (Innovation fond of the medical faculty of the Ulm University; successfully applied for by S.O. Reber)

Mechanisms underlying CSC-induced breakdown of HPA axis functions. (German Research Foundation, RE 2911/5-1; successfully applied for by S.O. Reber)

Effects of psychosocial stress on intestinal homeostasis and mechanisms underlying stress-induced colitis. (German Research Foundation, RE 2911/2-1; successfully applied for by S.O. Reber)
Research Profile of the Laboratory for Molecular Psychosomatics
To investigate our research hypotheses stated above, we employ several preclinically validated rodent models of chronic psychosocial stress and study in detail their effects on behavioral, physiological, neuroendocrine, and immunological parameters, thereby considering individual differences in the stress coping strategy. Our main paradigm in this context to induce chronic psychosocial stress during adulthood is the chronic subordinate colony housing (CSC) paradigm (Reber et al., 2007 PNEC; Reber, 2012 PNEC; Langgartner et al., 2015 Frontiers in Psychiatry; Reber et al., 2016 PNEC). The CSC model is based on repeated psychosocial traumatization (social defeat, Days 1, 8, and 15) and prolonged (19 d) social subordination of male experimental mice to a larger dominant male and results in many behavioral, endocrine and immunological changes known from posttraumatic stress disorders patients (for review see Reber et al., 2016 PNEC). The value and clinical relevance of this stress model has recently been acknowledged with the "Curt P. Richter Award" by the International Society for Psychoneuroendocrinology (ISPNE). To induce chronic psychosocial stress in mice during early life we employ the maternal separation (MS) paradigm (Veenema et al., 2008 Endocrinology). Here, the pubs are separated from the mother for 3h per day between postnatal days (PND) 1-14. MS negatively affects the behavior (i.e. increases anxiety-related behavior), alters HPA axis (re)activity, and promotes immune activation, which is in line with what has been reported in humans exposed to early life trauma/stress.
To induce acute psychosocial stress in human volunteers under standard laboratory conditions for investigating its effects on mood, physiology and the immune system, we employ a slightly modified version of the "Trier Social Stress Test (TSST)", originally described earlier (Kirschbaum et al., 1993 Neuropsychobiology). TSST exposure reliably activates the two main stress axes, namely the sympathetic nervous system (SNS) and the hypothalamus-pituitary-adrenal (HPA) axis, as well as the cardiovascular and the immune system.
Current Research Projects
P1
Immunoregulatory approaches to promote stress resilience
P2
Stress-induced glucocorticoid resistance
P3
Stress-induced bone defects
P4
Understanding the mechanisms underlying the increased prevalence of mental and physical stress-associated disorders in urban vs. rural areas.
For NEWS follow us on X: @MolPsySoUlm
News
- Ms. Tamara Schimmele received the PNIRS Asia-Pacific Scholar Award.
- Prof. Reber as guest at ARTE Saloon Live-Talk: "Können Mikroben alles?"
- The Reber Lab on TV - Doc Fischer at SWR: "Stress & Knochen"
- 2023 GEBIN Annual Meeting - the Reber MPS lab as local organizer
- Prof. Reber on YouTube - Blog "Immunsignature" by Dr. med. Keferstein (Mojo Institute for Regenerative Medicince)